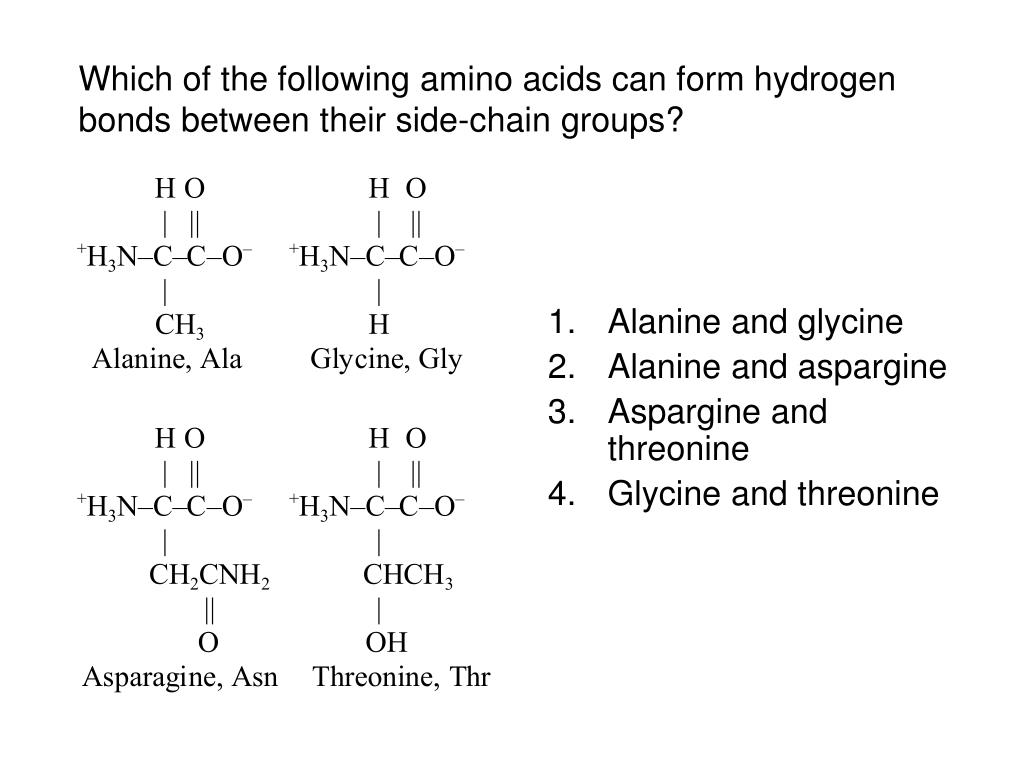
It is believed that proteins and tannins interact via hydrogen bonding and hydro-phobic effects. Hydrophilic proteins will elute first, whereas hydrophobic proteins elute last. After the protein mixture is applied to the column, an elution buffer with decreasing ionic strength is used. Hydrophobic interaction chromatography relies on hydrophobic interactions between apolar amino acid residues in the proteins and a resin containing hydrophobic groups, such as /7-octyl or phenyl groups. In contrast, charged amino acids are located almost exclusively on the surface of the molecule, where they can form hydrogen bonds, with each other and with water. They are packed closely together, forming a structure stabilized by hydrophobic interactions between these clustered residues (see p. Location of polar and nonpolar amino acid residues The interior of the myoglobin molecule is composed almost entirely of nonpolar amino acids. Hydrophobic interactions between amino acids with nonpolar side chains. Integral proteins associate firmly with membranes by hydrophobic interactions between the lipid bilayer and their nonpolar amino acid side chains, which are oriented toward the outside of the protein molecule. Peripheral proteins are loosely associated with the membrane through electrostatic interactions and hydrogen bonds or by covalently attached lipid anchors. It displays a tyrosine kinase domain, which includes several tyrosine autophosphorylation sites, and an actin-binding domain that may facilitate interaction with the cell cytoskeleton. The receptor cytoplasmic region contains some 542 amino acids. A 23 amino acid hydrophobic domain spans the plasma membrane.
It displays two cysteine-rich regions, between which the ligand-binding domain is located. The N-terminal, extracellular region of the receptor contains 622 amino acids. Common conformations include a-helices and P pleated sheets (see Chapter 5). The physiologically active conformation reflects the amino acid sequence, steric hindrance, and noncovalent interactions (eg, hydrogen bonding, hydrophobic interactions) between residues. Amino acids hydrophobic interactions betweenįolding of a peptide probably occurs coincident with its biosynthesis (see Chapter 38).
